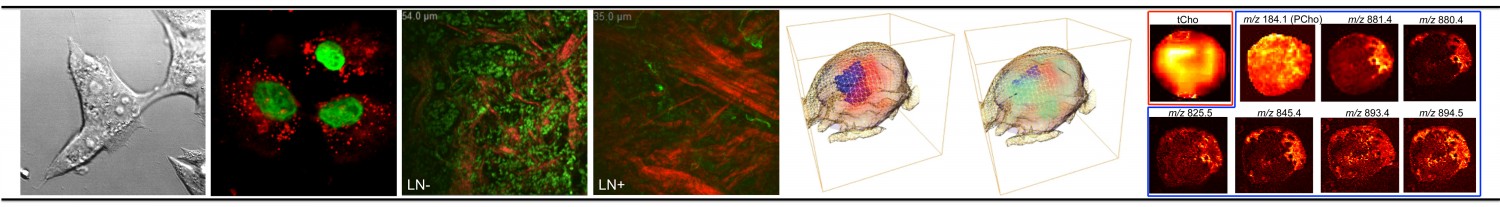
Abnormal choline metabolism is emerging as a metabolic hallmark that is associated with oncogenesis and tumor progression. Following transformation, the modulation of enzymes that control anabolic and catabolic pathways causes increased levels of choline-containing precursors and breakdown products of membrane phospholipids. These increased levels are associated with proliferation, and recent studies emphasize the complex reciprocal interactions between oncogenic signaling and choline metabolism. Because choline-containing compounds are detected by non-invasive magnetic resonance spectroscopy (MRS), increased levels of these compounds provide a non-invasive biomarker of transformation, staging and response to therapy. Furthermore, enzymes of choline metabolism, such as choline kinase, present novel targets for image-guided cancer therapy. For additional reading please go to Glunde et al, Nature Reviews Cancer, 2011.
Molecular Studies of the MR-detectable Oncometabolite Glycerophosphocholine (R01CA213428)
Choline phospholipid metabolism is profoundly altered in cancer. While a lot of research has been devoted toward elucidating the molecular origins of elevated phosphocholine (PC) in cancer, little attention has been paid to the oncometabolite glycerophosphocholine (GPC) and its underlying molecular biology. High-resolution (HR) magnetic resonance spectroscopy (MRS) studies have shown that GPC is higher than PC in normal breast epithelial cells, whereas PC is higher than GPC in breast cancer cells. This finding is known as “GPC to PC switch” in the choline metabolite profile upon malignant transformation. To date, the genes and specific enzymes responsible for the cancer-related alterations in GPC are unknown. The changes in cellular GPC levels could occur through glycerophosphocholine-phosphodiesterase (GPC-PDE) or lysophospholipase (LPL). By combining HR MRS with gene silencing strategies, we have observed that silencing of GDPD5 and GDPD6, both of which are GPC-PDEs, significantly increased the GPC levels in breast cancer cells, while reducing their proliferation, migration, and invasion. Chemotherapy treatment of breast cancer cells with doxorubicin resulted in down-modulation of the choline cycle genes choline kinase alpha (Chkα), phospholipase D1 (PLD1), and GDPD6, thereby resulting in a net increase of GPC and a decrease in PC along with a significant inhibition of proliferation. These preliminary data clearly point towards an important role of GDPD5 and GDPD6 in breast cancer cell aggressiveness and treatment response to chemotherapy. Our new preliminary data with three different commonly used FDA-approved chemotherapeutic agents indicate that there is value in exploring GPC as an additional biomarker of treatment response independent of PC, which, of course, would be in addition to other valuable imaging biomarkers. We will also investigate the reciprocal interactions between oncogenic signaling pathways and GPC-regulating enzymes that are critical for tumor maintenance and progression. The proposed research will provide novel insight into the molecular regulation of the oncometabolite GPC in breast cancer, and will help understand its role in cancer progression. Our findings will provide the foundation for interpreting GPC levels detected by noninvasive 1H and 31P MRS as an additional complementary biomarker of transformation, staging, and response to therapy. GPC or critical GPC- regulating enzymes may also be useful biomarkers of transformation, staging, and response to therapy in biopsied tissue samples ex vivo. The enzymes studied in this proposal, e.g. GDPD5 and GDPD6, may prove useful as anticancer targets, either alone or in combination with chemotherapy treatment of breast cancer.